Say goodbye to low-lying eyelids. Our Atlanta and Alpharetta offices now offers Upneeq. An FDA-approved prescription eyedrop for acquired blepharoptosis (also known as acquired ptosis or low-lying lids) in adults, proven to rapidly lift the upper eyelid.1,4.
What is Acquired Blepharoptosis?
Acquired ptosis (low-lying lids) is a common eyelid disorder that can impact adults of all ages. It can make eyes appear tired/sleepy/droopy or make on eyelid visibly lower than the other. And may cause vision impairment, interfering with day to day functions such as reading, driving and computer use.
If your eyes look “tired” or “sleepy” schedule an appointment to see if Upneeq is the right treatment for you.
Contact Us For a Consultation
For all other med spa procedures please consider scheduling directly online.
What Can Upneeq® Do?
- Lifts droopy eyelids
- Improves field of vision, which may help you see better. Upneeq demonstrated 3 times greater improvement in visual field compared to vehicle in clinical trials (P<.01), as measured by LPFT (on Day 1 and Day 14 of treatment)1
- A daily drop of Upneeq delivers rapid, visible lift to the upper eyelid by stimulating the Müller’s muscle to raise the upper eyelid
Upneeq® Results After One Dose
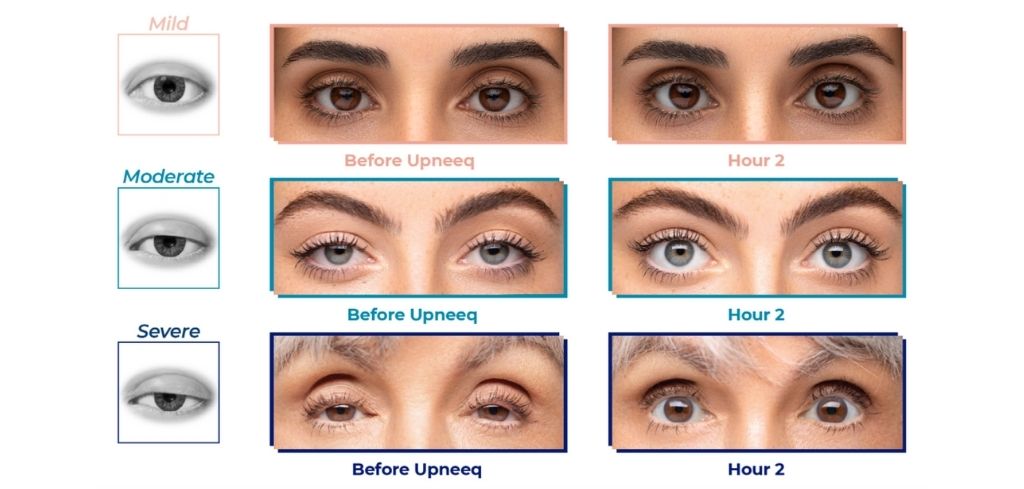
Am I a Good Candidates for Upneeq®
If you have this problem and want to correct it to improve vision and your overall look, you aren’t taking any medications that will interfere and you are in otherwise good health, you could be an ideal candidate for Upneeq. Dr. Rudderman can determine this during your consultation. Schedule a consultation at our Midtown Atlanta or Alpharetta locations.
LPFT=Leicester Peripheral Field Test (measuring superior visual field).
*Two randomized, multicenter, double-masked, vehicle-controlled, Phase 3 studies compared once-daily Upneeq in 304 subjects with acquired blepharoptosis. Efficacy was assessed with the Leicester Peripheral Field Test (LPFT) and photographic measurement of Marginal Reflex Distance (MRD-1). There was a statistically significant difference in mean change in the LPFT from baseline after instillation of Upneeq and vehicle, with significantly greater increases in the study eye of the Upneeq group evident at the 2-hour point and maintained at the 6-hour time point. Greater MRD-1 increases were observed for the Upneeq group than the vehicle group on Day 1 at 6 hours post-dose and on Day 14 at 2 hours post-dose.1
INDICATION
UPNEEQ® (oxymetazoline hydrochloride ophthalmic solution), 0.1% is indicated for the treatment of acquired blepharoptosis in adults.
IMPORTANT SAFETY INFORMATION
WARNINGS AND PRECAUTIONS
- Ptosis may be associated with neurologic or orbital diseases such as stroke and/or cerebral aneurysm, Horner syndrome, myasthenia gravis, external ophthalmoplegia, orbital infection and orbital masses. Consideration should be given to these conditions in the presence of ptosis with decreased levator muscle function and/or other neurologic signs.
- Alpha-adrenergic agonists as a class may impact blood pressure. Advise UPNEEQ patients with cardiovascular disease, orthostatic hypotension, and/or uncontrolled hypertension or hypotension to seek medical care if their condition worsens.
- Use UPNEEQ with caution in patients with cerebral or coronary insufficiency or Sjögren’s syndrome. Advise patients to seek medical care if signs and symptoms of potentiation of vascular insufficiency develop.
- UPNEEQ may increase the risk of angle closure glaucoma in patients with untreated narrow-angle glaucoma. Advise patients to seek immediate medical care if signs and symptoms of acute narrow-angle glaucoma develop.
- Patients should not touch the tip of the single patient-use container to their eye or to any surface, in order to avoid eye injury or contamination of the solution.
ADVERSE REACTIONS
Adverse reactions that occurred in 1-5% of subjects treated with UPNEEQ were punctate keratitis, conjunctival hyperemia, dry eye, blurred vision, instillation site pain, eye irritation and headache.
DRUG INTERACTIONS
- Alpha-adrenergic agonists, as a class, may impact blood pressure. Caution in using drugs such as beta blockers, anti-hypertensives, and/or cardiac glycosides is advised. Caution should also be exercised in patients receiving alpha adrenergic receptor antagonists such as in the treatment of cardiovascular disease, or benign prostatic hypertrophy.
- Caution is advised in patients taking monoamine oxidase inhibitors which can affect the metabolism and uptake of circulating amines.
To report SUSPECTED ADVERSE REACTIONS or product complaints, contact RVL Pharmaceuticals at 1-877-482-3788. You may also report SUSPECTED ADVERSE REACTIONS to the FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
References: 1. Upneeq® (oxymetazoline hydrochloride ophthalmic solution) 0.1%. [Prescribing Information]. RVL Pharmaceuticals, Inc. 2020. 2. Finsterer J. Ptosis: causes, presentation, and management. Aesthetic Plast Surg. 2003;27(3):193-204. 3. Richards HS, Jenkinson E, Rumsey N,et al. The psychological well- being and appearance concerns of patients presenting with ptosis. Eye. 2014; 28(3):296-302. 4. Data on file, RVL Pharmaceuticals, Inc
